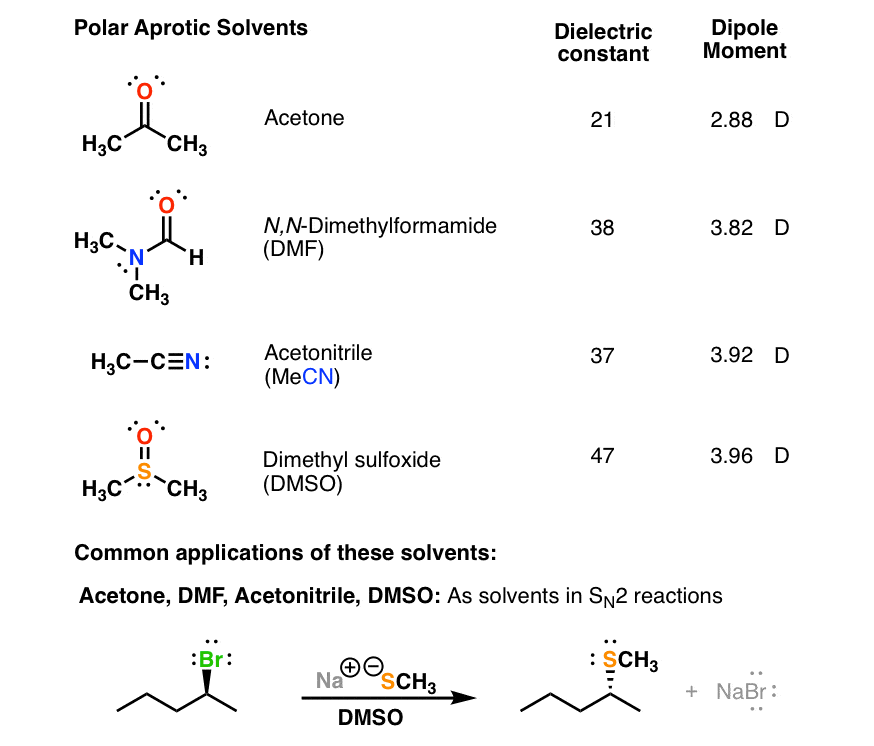
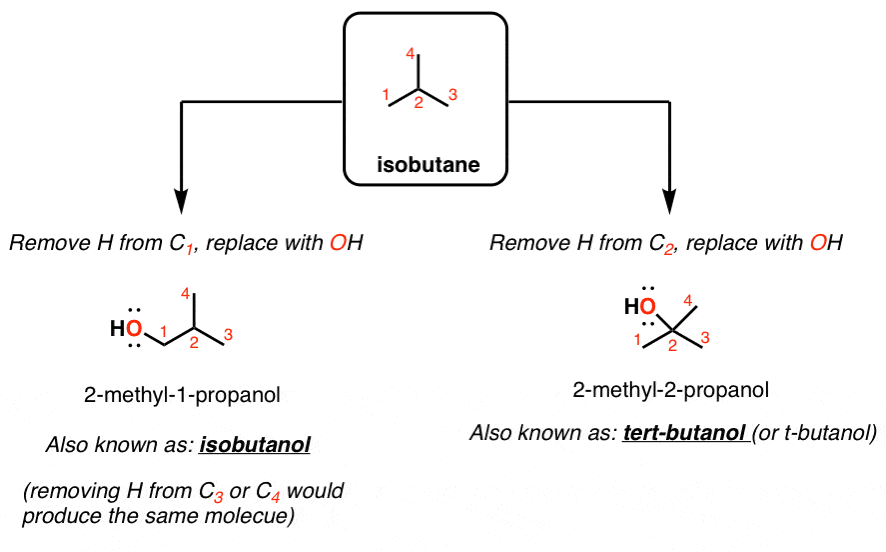
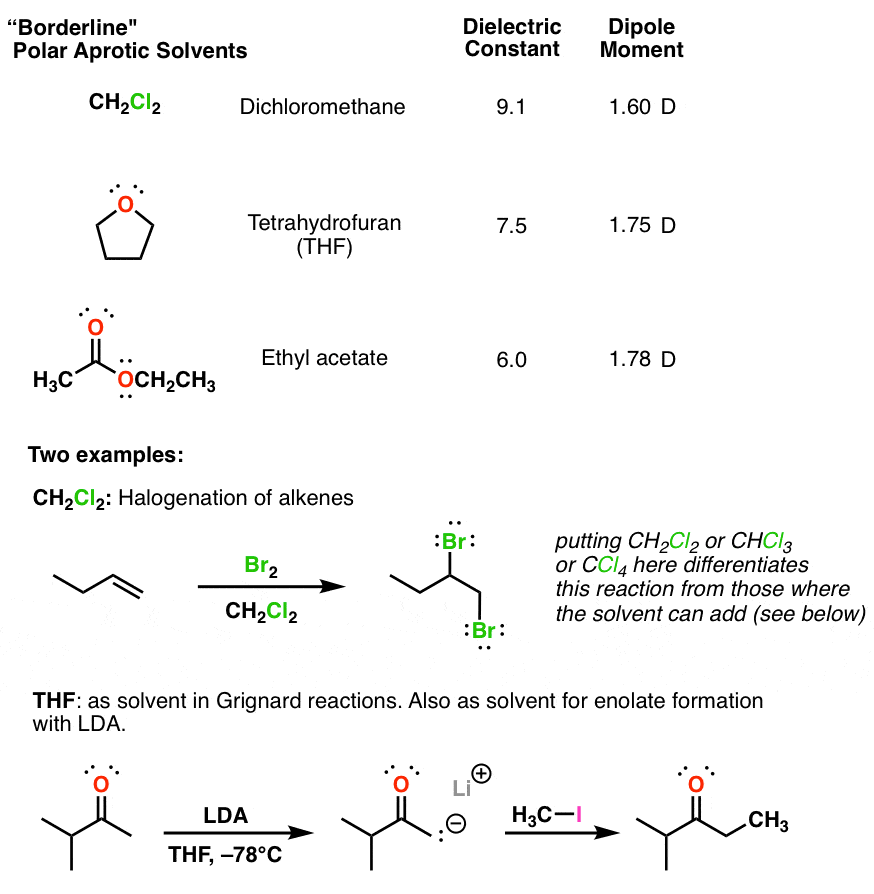
Arrange the following solvents in order of increasing polarity: a) ethanol b) ethyl acetate c) petroleum ether d) toluene e) acetone | Homework.Study.com

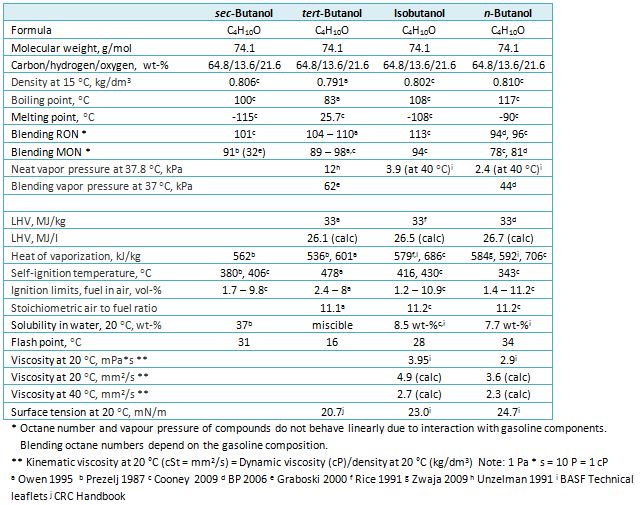
Thermodynamic properties and structure transition in {water + tert-butanol} and {water + tert-butanol + iso-butanol} solutions - ScienceDirect

SOLVED: When t-butyl methyl ether is treated with one equivalent of anhydrous HI in ether (a nonpolar solvent) , t-butanol and methyl lodide are formed: (CHs)C-O-CHs (CHb)C-OH CHsl When the reaction performed

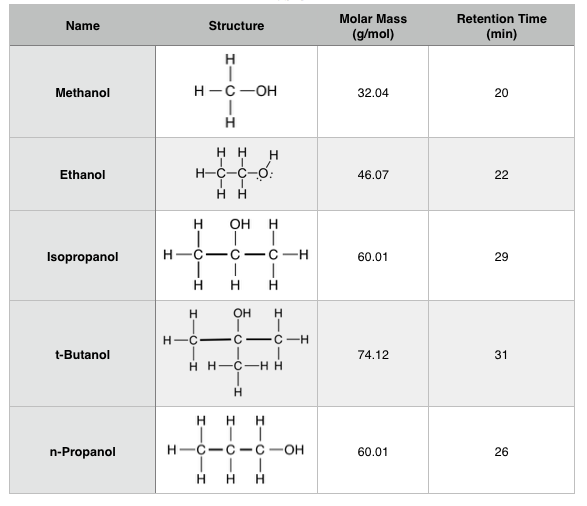
SOLVED: Experimental condition (Fig: and Fig.2: GC column: RTF-5 containing non-polar C18 polydimethyl siloxane stationary phase. This column can well separate the -components mixture except the overlapping of one ingredient with solvent

Rationalizing the Mechanism of Peroxyformate Decomposition: Computational Insights To Understand Solvent Influence - Garay‐Ruiz - 2021 - Chemistry – A European Journal - Wiley Online Library